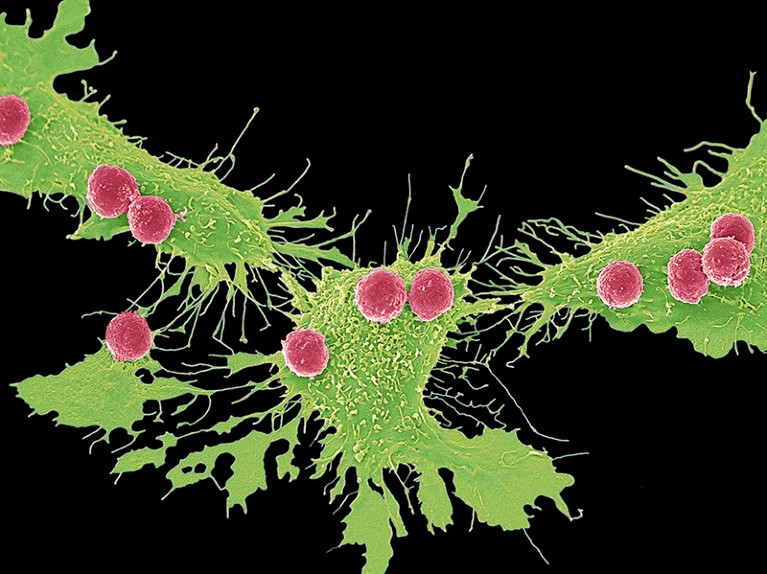
Engineered cells called CAR T cells (pictured, red; artificially coloured) swarm cancer cells (green). A tool that edits RNA can restore the efficiency of ‘exhausted’ CAR T cells.Credit: Steve Gschmeissner/Science Photo Library
The CRISPR–Cas9 gene-editing system excels at altering and disrupting genes. But the changes it makes are permanent, which can be a big problem if the system goes awry. Now, a CRISPR-based system that targets a cell’s short-lived messenger RNA instead of DNA could provide a more precise and reversible way of designing cell therapies — and even help scientists to discover how different genes work together.
The results were published on 21 February in Cell1.
RNA gets its turn
Table of Contents
Engineered CRISPR systems generally have two main components: a DNA-cutting enzyme, often Cas9, and a piece of ‘guide’ RNA that directs the enzyme to the stretch of DNA to be edited. One of the system’s most promising medical applications has been its potential use in producing chimeric antigen receptor (CAR) T cells. These are made by engineering the immune foot soldiers called T cells to attack specific proteins on the surfaces of tumour cells. But DNA-editing CRISPR systems can pose safety problems and are relatively inefficient in these cells.
CRISPR 2.0: a new wave of gene editors heads for clinical trials
Bioengineer Stanley Qi and immunologist Crystal Mackall, both at Stanford University in California, and their colleagues developed an alternative system, called MEGA (multiplexed effector guide arrays). It has CRISPR guide RNA but swaps the DNA-cutting Cas9 for an RNA-cutting alternative called Cas13d. The CRISPR half of the duo directs Cas13d to a target mRNA, which is produced from a DNA template.
“We are not really touching any DNA,” Qi says. This avoids the risk of inducing permanent changes or, worse, cutting DNA in places other than the designated target. The mRNA doesn’t last very long in a cell, so any mistakes will quickly disappear.
Active cells such as T cells produce a constantly changing variety of mRNA molecules, each directing the production of a specific protein. Cas13d cuts the target mRNA, destroying it and preventing it from churning out its specific protein. This has the same effect as turning off the associated gene. MEGA allowed the researchers to create ‘multiplex’ CRISPR–Cas13d systems that can shut down the production of multiple proteins, effectively turning off up to ten genes at a time.
Rejuvenating exhausted cells
The team used the system to address a shortcoming of CAR-T therapy called T-cell exhaustion. If CAR T cells are activated too many times by a chronic infection or a long-term tumour, they become less effective.
To give a jolt to tired T cells, the researchers designed CRISPR systems that target mRNA molecules involved in functions including energy production and sugar metabolism. T cells treated with some MEGA combinations stopped expressing molecular signals of exhaustion and became better at shrinking tumours in mice.

The race to supercharge cancer-fighting T cells
Qi, Mackall and their colleagues also created a version of Cas13d that is switched on only when the CAR T cells are treated with the antibiotic trimethoprim. By varying the doses of trimethoprim, the researchers could ‘tune’ mRNA levels up and down, giving the team precise control over when and how molecular pathways were activated, rather than just shutting them down entirely.
“It’s always thrilling to see how the RNA CRISPR toolbox is applied,” says systems biologist Jonathan Gootenberg at the Massachusetts Institute of Technology in Cambridge. The ability to tune the collection of RNA transcripts, he says, will be especially useful for cell therapies.
Joseph Fraietta, an immunologist at the University of Pennsylvania in Philadelphia, agrees. In his own experience with CRISPR, he says, his group can edit only about three genes in CAR T cells at a time before the cells become unhealthy. “This will open more avenues,” he says. But he cautions that the system requires continuously high levels of Cas13d, which might trigger an immune response.
Mackall and Qi say that MEGA’s ability to tune gene expression allows scientists to vary the levels of a wide array of mRNAs at one time, revealing how different amounts of mRNA from various combinations of genes work together to carry out cellular functions.
