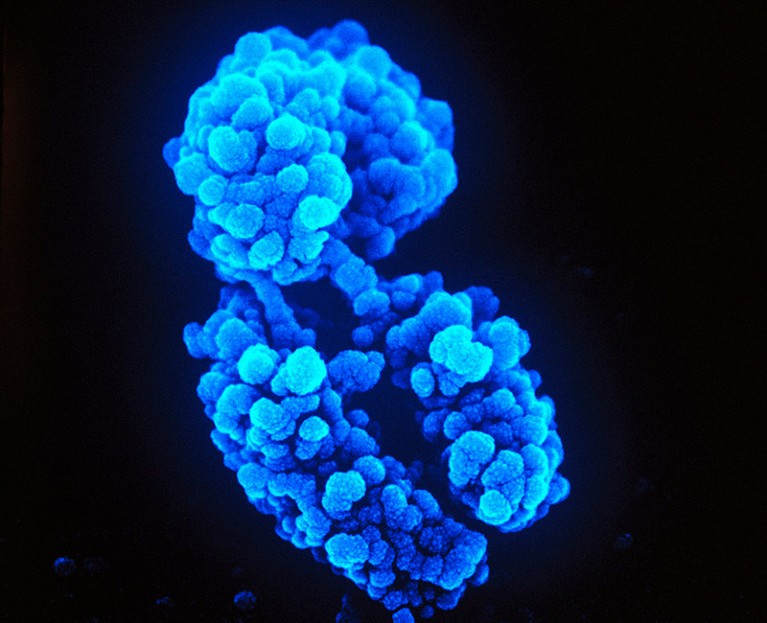
Protein–RNA complexes that shroud some copies of the X chromosome (artificially coloured) contribute to the female bias in prevalence of autoimmune disease.Credit: Lennart Nilsson, TT/Science Photo Library
Why are women so much more susceptible to autoimmune diseases than men? A new explanation for the discrepancy has emerged: a molecular coating typically found on half of a woman’s X chromosomes — but not in males’ cells — might be provoking unwanted immune responses1.
The coating, a mix of RNA and proteins, is central to a developmental process called X-chromosome inactivation. Researchers had previously implicated sex hormones and flawed gene regulation on the X chromosome as drivers of the autoimmune disparity. But the discovery that proteins central to X-chromosome inactivation can themselves set off immunological alarm bells adds yet another layer of complexity — and could point to new diagnostic and therapeutic opportunities.
“This really adds a new mechanistic twist,” says Laura Carrel, a geneticist at the Pennsylvania State College of Medicine in Hershey.
The study was published today in Cell1.
Medical mystery
Table of Contents
Women account for around 80% of all cases of autoimmune disease, a category that includes conditions such as lupus and rheumatoid arthritis. What explains this sex bias has long been a mystery, however.
“It’s a question that’s been irking immunologists and rheumatologists for the past 60 or 70 years,” says Robert Lahita, a rheumatologist at the Hackensack Meridian School of Medicine in Nutley, New Jersey.
Ancient DNA reveals first known case of sex-development disorder
A prime suspect is the X chromosome: in most mammals, including humans, a male’s cells typically include only one copy, whereas a female’s cells typically carry two.
(This article uses ‘women’ and ‘female’ to describe people with two X chromosomes and no Y chromosome, reflecting the language of the study, while acknowledging that gender identity and chromosomal make-up do not always align.)
X-chromosome inactivation muffles the activity of one X chromosome in most XX cells, making their ‘dose’ of X-linked genes equal to that of the XY cells typical in males. The process is highly physical: long strands of RNA known as XIST (pronounced ‘exist’) coil around the chromosome, attracting dozens of proteins to form complexes that effectively muzzle the genes inside.
Not all genes stay mum, however, and those that escape X inactivation are thought to underpin some autoimmune conditions. Additionally, the XIST molecule itself can initiate inflammatory immune responses, researchers reported in 20232. But that is not the whole story.
XISTential questions
Almost a decade ago, Howard Chang, a dermatologist and molecular geneticist at Stanford University School of Medicine in California and a co-author of the current study, noticed that many of the proteins that interact with XIST were targets of misguided immune molecules called autoantibodies.
‘It’s all gone’: CAR-T therapy forces autoimmune diseases into remission
These rogue actors can attack tissues and organs, leading to the chronic inflammation and damage characteristic of autoimmune diseases. Because XIST is normally expressed only in XX cells, it seemed logical to think that the autoantibodies that attack XIST-associated proteins might be a bigger problem for women than for men.
To test this idea, Chang and his colleagues turned to male mice, which don’t usually express XIST. The team bioengineered the mice to produce a form of XIST that did not silence gene expression but did form the characteristic RNA–protein complexes.
The team induced a lupus-like disease in the mice and found that animals that expressed XIST had higher autoantibody levels than those that didn’t. Their immune cells were also on higher alert, a sign of predisposition to autoimmune attacks, and they showed more extensive tissue damage.
Immune-system overdrive
Notably, the same autoantibodies were also identified in blood samples from people with lupus, scleroderma and dermatomyositis — evidence that XIST and its associated proteins are “something that our immune systems have trouble ignoring”, says Allison Billi, a dermatologist at the University of Michigan Medical School in Ann Arbor.
Montserrat Anguera, a geneticist at the University of Pennsylvania in Philadelphia, points to the human data as validation that the XIST-related mechanisms observed in mice have direct relevance to human autoimmune conditions, with implications for disease management. For example, diagnostics targeting these autoantibodies could assist clinicians in detecting and monitoring various autoimmune disorders.
“This is a cool start,” she says. “If we could use this information to expedite the diagnosis, it would be amazing.”
