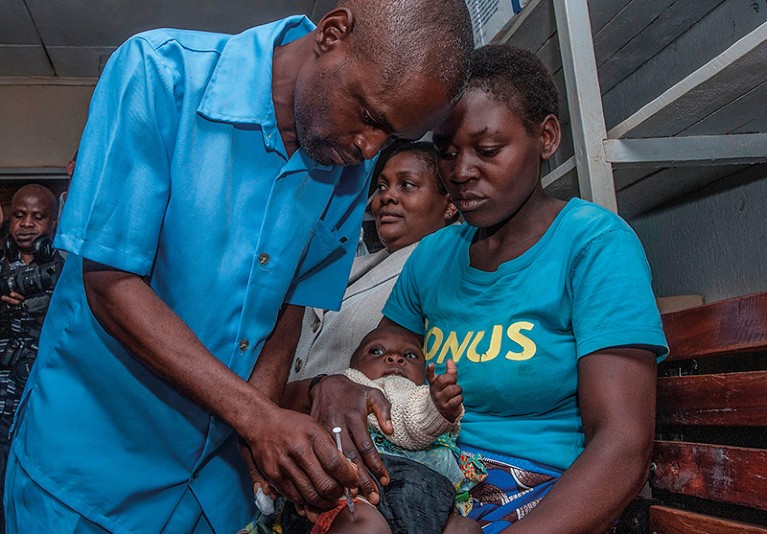
Ghana, Kenya and Malawi took part in a pilot study of the RTS,S vaccine in 2019.Credit: Amos Gumulira/AFP via Getty
John Bawa, who leads vaccine implementation in Africa at the global non-profit organization PATH in Accra, has been working for more than a decade on the first vaccine against malaria. And he has become used to hearing the same question: “Where is your vaccine?” So, last year, when the World Health Organization (WHO) recommended the use of the vaccine, known as RTS,S and marketed as Mosquirix, in children living in countries hardest hit by the disease, “it was a great relief for us”, he says. “Now I have my vaccine.”
The WHO’s recommendation was a historic milestone. RTS,S took 30 years to develop, and is not only the first malaria vaccine, but also the first vaccine for any parasitic disease. Although the efficacy of the shot is modest — about 50% in the first year1 — it is expected to save tens of thousands of lives each year. One study2 estimated that, if the vaccine were rolled out in countries with the highest burden of malaria, it could prevent 5.3 million cases and 24,000 deaths in young children each year.

Part of Nature Outlook: Children‘s health
But reaping that benefit will take time. So far, more than one million children have received one or more doses of the vaccine in a pilot study in Ghana, Kenya and Malawi. That’s just a fraction of the 25 million children in more than 30 countries who need it. It could be years before many of those countries get their first doses.
Specialists estimate that demand will be 80 million to 100 million doses per year. The vaccine’s manufacturer GlaxoSmithKline (GSK), a pharmaceutical company based in Brentford, UK, has promised to deliver 18 million doses over the next 3 years. For the people who have watched this vaccine hit roadblock after roadblock during its long period of development, the supply problems come as a disappointment.
But Dyann Wirth, a malaria researcher at the Harvard T.H. Chan School of Public Health in Boston, Massachusetts, is trying to look on the bright side. Having some vaccine is much better than having none. RTS,S approval “changed the conversation about whether vaccines were feasible for malaria”, she says. And that will pave the way for better vaccines.
The long delay
Table of Contents
Public-health workers have made impressive gains against malaria in recent decades. Between 2000 and 2019, global deaths attributable to malaria fell by around 40%. Yet the disease remains one of the top causes of death among children. In 2020, the world saw 241 million cases of malaria and 627,000 deaths. Africa accounted for around 95% of the cases and deaths, and 80% of those deaths were children under 5.
RTS,S works by targeting a portion of the circumsporozoite protein on the surface of the malaria parasite. The idea is that a vaccinated individual will generate antibodies and kill off the parasite before it can enter red blood cells. But the vaccine does not grant perfect protection. In the first year after vaccination, it is about 50% effective in preventing clinical cases of malaria in children aged 5 to 17 months1. After 4 years, efficacy drops to 36% for clinical episodes and 32% for severe malaria. The vaccine has also been tested in younger infants aged between 6 and 12 weeks, but efficacy was lower and the benefits were not deemed significant enough to justify its use in this age group.
Those efficacy numbers are disappointing when compared with vaccines for measles or polio, which are more than 90% protective. But “when we put that in the spectrum of how effective our other interventions are at preventing severe malaria, it’s not as bad as it seems”, says Joshua Yukich, an epidemiologist at Tulane University in New Orleans, Louisiana. “It’s not like giving a person a bed net prevents 100% of severe malaria episodes.” And the hope is that the vaccine will be added to other interventions that are already in place. In fact, when researchers combined the vaccine with medications to prevent malaria in areas with seasonal transmission, the combination offered much higher protection against the disease than either strategy alone3 — around 60%.
The phase III trial for RTS,S wrapped up in 2014. For most vaccines, this is the final step before approval and roll-out. But the vaccine needed to pass the WHO’s prequalification programme, which certifies the safety and efficacy of medicines and vaccines destined for low- and middle-income countries. When the WHO’s advisory committee on immunization and malaria met to discuss the vaccine, some members had concerns.
The study had not been able to determine the impact of RTS,S on mortality. What’s more, researchers had picked up cases of meningitis in children who had received the vaccine, and the WHO committee members wanted to be sure there wasn’t a causal link. They also questioned the feasibility of rolling out a four-dose vaccine in Africa. The timing of the doses doesn’t entirely line up with those for other childhood vaccines, and reaching children after their first year of life can be tricky. “People were very sceptical that an African health-care system was going to be able to deliver that vaccine in a way that was worth it,” Yukich says.
So rather than giving RTS,S a full recommendation, the WHO decided to green-light a large-scale pilot study in 2019 in Ghana, Kenya and Malawi. That study doesn’t officially wrap up until 2023, but by October 2021 the WHO and its advisers had enough data to see that the vaccine was safe, uptake was good and implementation looked feasible (see go.nature.com/3ilss7v).
Running a pilot study made sense at the time, Wirth says — this was the first vaccine for parasitic disease ever used in people, after all. But she says it’s fair to question whether the right balance was struck between safety and urgency. “Data from the phase II trials is more or less the same as the data from the pilot studies,” she says. The phase II results were published more than a decade ago. During that period “hundreds of thousands of children suffered serious malaria, and many of them died”.
Roll-out woes
The WHO’s endorsement is an important step in getting RTS,S to the children who need it, but most of those children won’t benefit any time soon. When the WHO initially decided to launch the pilot study in 2015, GSK closed their RTS,S manufacturing plant in Wavre, Belgium. It didn’t reopen until 2019, and ramping up production will take time.
GSK has promised to deliver 4 million doses in 2023, 6 million in 2024 and 8 million in 2025. By 2026, GSK’s plant will produce 15 million doses a year. “That is all the facility can produce,” says Thomas Breuer, chief global health officer at GSK. But that number is a fraction of the expected demand.
“There are 40 million kids born in sub-Saharan Africa alone in malarious areas of moderate to high transmission,” says Adrian Hill, director of the Jenner Institute at the University of Oxford, UK, and RTS,S requires four doses. “That’s 160 million doses a year.” The WHO predicts that demand will be lower — in the realm of 100 million doses per year. But both estimates represent much more vaccine than GSK plans to produce.
GSK has announced that, by 2028, it will transfer the technology to manufacture RTS,S to Bharat Biotech, a biotechnology company in Hyderabad, India. This should help to bolster supplies, but there’s a catch. RTS,S is composed of two parts: the antigen (RTS,S) and an adjuvant called AS01E, which helps to boost the immune response. Bharat Biotech will manufacture the antigen, but GSK will still supply the adjuvant. AS01E contains a type of chemical called a saponin — specifically, “a particular saponin you can only extract from a tree called Quillaja saponaria, which grows primarily in Chile”, Hill says. GSK has promised to supply up to 30 million doses of adjuvant per year. “If more is needed, we will find it,” Breuer says. He points out that the vast majority of this saponin is used for other purposes, such as in cosmetics, and could be diverted. “The adjuvant will not be the bottleneck,” he says.
Until then, however, the limited supplies of vaccine that do exist will have to be carefully distributed. That task falls to Gavi, the Vaccine Alliance, in Geneva, Switzerland, which helps to vaccinate children in the world’s poorest countries. In December 2021, Gavi’s board approved US$155.7 million for the roll-out of RTS,S from 2022 to 2025. Countries that want the vaccine can apply for funding from Gavi, which will consider a variety of factors, including each nation’s malaria burden and capacity to deploy the vaccine. Countries that receive vaccines through Gavi will pay a variable portion of the cost, depending on their income.
Only the three countries that piloted the vaccine were eligible for the first round of funding. “We want to make sure that we can get sufficient supplies to them so that they can continue their programmes without having any pause,” says Stephen Sosler, head of vaccine programmes at Gavi. The next round will close in January 2023. As of late November, 21 countries had expressed interest in applying. Gavi is working hard to try to get those countries their first doses by the end of 2023. Because supplies are so limited, Gavi will take extra care to make sure that each dose has the maximum impact and that there isn’t any waste, Sosler says.
Alternatives on trial
Having multiple vaccines would help to ease supply constraints. In July, the biotechnology company BioNTech, based in Mainz, Germany, announced plans to launch trials of an mRNA vaccine for malaria by the end of this year. Another malaria vaccine, being developed at the University of Oxford, called R21, is already in phase III clinical trials and, if proven to be safe and effective, it could be deployed as soon as 2023. R21 uses the same antigen as RTS,S, but the results of R21’s phase II trials, released in September4, suggest that it might be more effective. In a trial of about 400 children in Burkina Faso, 4 doses provided around 75% protection against clinical cases of malaria after 12 months.

More from Nature Outlooks
That number sounds impressive, but whether it will hold up in the phase III trial isn’t clear. The phase II study occurred in West Africa, where malaria transmission is seasonal. “There’s a peak of malaria in September, October and November. We were vaccinating in June and July,” says Hill, just before the malaria season hit. But, “what will happen when you go to a place where transmission is over 12 months of the year?” says Halidou Tinto, the regional director at the Institute of Research in Health Sciences (IRSS) in Nanoro, Burkina Faso.
If R21 is approved, it could drastically boost malaria vaccine supplies. The world’s largest vaccine manufacturer — the biopharmaceuticals company Serum Institute of India in Pune — has already committed to producing more than 200 million doses per year, a quantity that Tinto calls “amazing”.
Of course, getting any malaria vaccine to low-income countries will require a hefty and sustained investment. “We hoped that the kind of support that COVID had, with all the funding, with all the interest, would have come on board,” Bawa says. That hasn’t happened, yet. But he still hopes it will. “The fight against malaria is a global fight,” he says. If the world can eliminate malaria, “posterity would never forget us”.
